HS - Chemistry
C.1 Thermodynamics in Earth’s Systems
13 lessons | 30 days of instruction
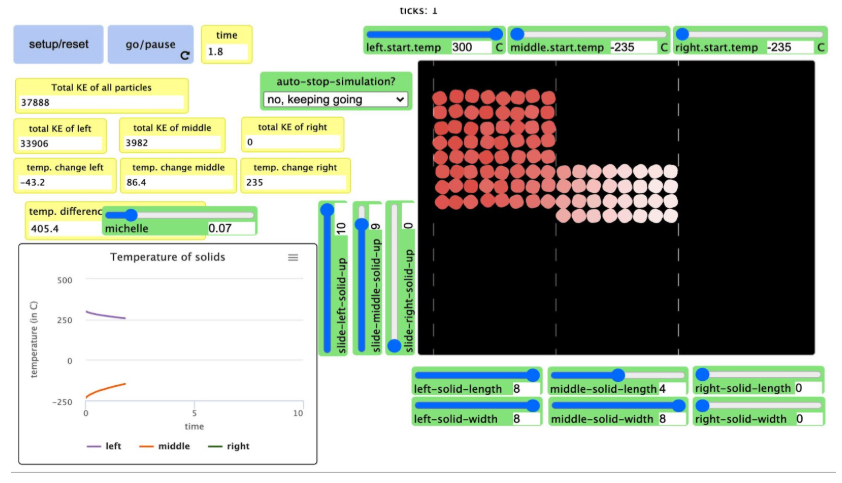
Why is the sea level rising, causing some people to have to move? Initial student models in this unit propose a variety of ideas, but it seems like melting polar ice is a likely cause for this global phenomenon. Uncertainty and student concern for the people impacted motivate unit investigations that help students better understand the matter and energy flows that underlie a global phenomenon like polar ice melt and sea level rise.
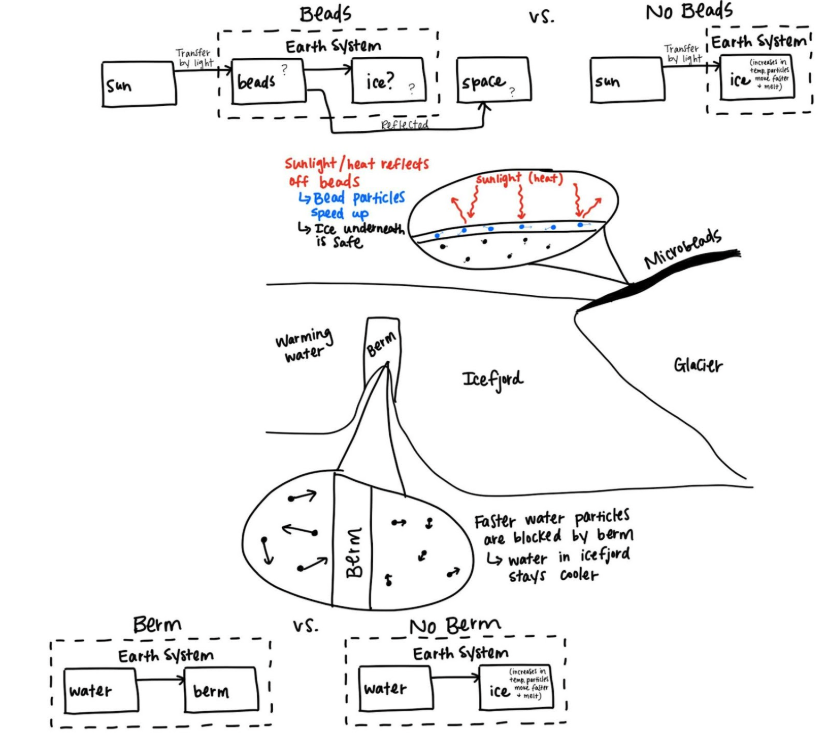
Historical data, hands-on investigations, and typical early-year math (like unit conversions) help students establish the mechanisms that cause sea level rise and estimate its potential impact. Through investigations, simulations, and system models, students figure out how decreasing carbon dioxide emissions and two geoengineering solutions (applying glass microbeads to polar ice and protecting glaciers from warm water with berms) could help slow polar ice melt, protecting coastal communities. As they do so, they…
begin developing the science practices needed in a chemistry classroom,
build a particle-level, quantifiable understanding of thermodynamics, and
consider how human activity results in particle-level changes with global implications.
Learn more in the Unit Storyline
SINGLE KIT | $
All materials for 1 class
MULTI KIT | $
Materials for 6 sections
ADD-ON | $
Consumables for 1 class
C.2 Structure & Properties of Matter
14 lessons | 29 days of instruction
What causes lightning and why are some places safer than others when it strikes? This unit is designed to help students build a deeper understanding of atomic structure and atomic-scale force interactions through exploration of phenomena surrounding lightning and other static interactions. Students engage with stories and data about lightning and investigate a similar phenomenon in water droppers. They further investigate static interactions with various materials, including sticky tape, digging down to the subatomic level. Students apply these ideas back to lightning and further investigate force interactions, developing Coulomb’s law and ideas about polarization that can be applied to other phenomena. They identify electric fields as the source of the large energy transfers in lightning and explain lightning’s sudden behavior using ionization. They consider why structures made of certain materials provide protection from lightning and investigate why bodies of water, most of which contain dissolved salts, are particularly dangerous during storms. Finally, students develop a consensus model and transfer their understandings to the phenomena of airplane radomes and conducting gels used to simulate brains.
Learn more in the Unit Storyline
SINGLE KIT | $
All materials for 1 class
MULTI KIT | $
Materials for 6 sections
ADD-ON | $
Consumables for 1 class
C.3 Molecular Processes in Earth Systems
15 lessons | 31 days of instruction
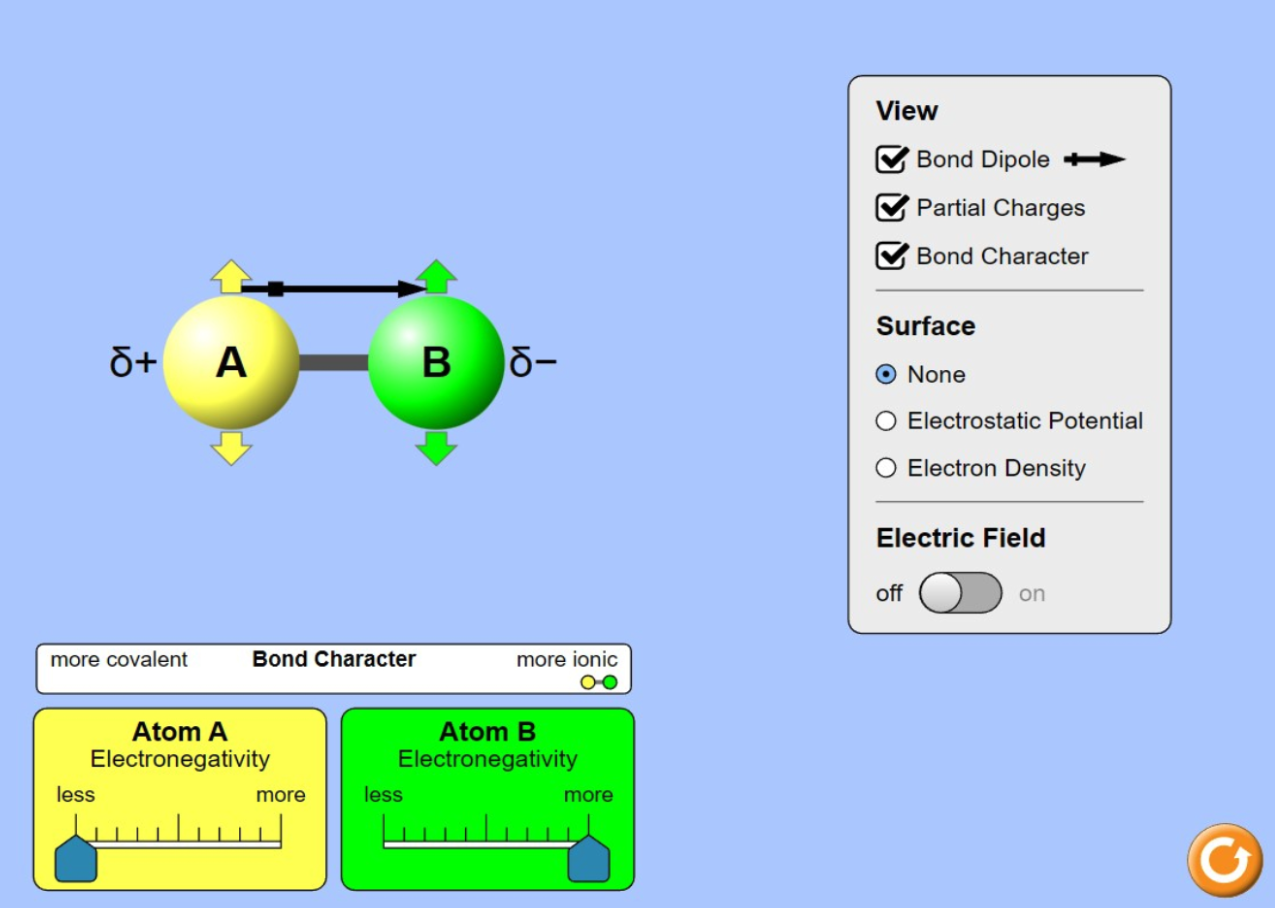
This unit is designed to deepen student understanding of atomic structure, trends on the periodic table, and how atomic-level interactions influence bulk-scale properties. The unit focuses on what substances we would need to find, make, and recycle in order to successfully live and work beyond Earth, or in space. Lesson Set 1 has students investigate properties of different liquids and their interactions with surface materials. Students use this information to predict which liquid or event resulted in a land formation on Earth, the Moon, or Mars. Lesson set 2 has students fully develop atomic structure and use patterns in how different elements interact with each other to build out the periodic table. Students then are able to relate bulk-scale properties to atomic-scale interactions, like electronegativity. Students continue to connect the atomic- and bulk-scale properties in Lesson Set 3. They use the law of conservation of matter as they balance chemical equations. In Lesson Set 4, students focus on how we can recycle substances by investigating how differences in the structures of the substance affect our ability to recycle it into different substances.
Learn more in the Unit Storyline
SINGLE KIT | $
All materials for 1 class
MULTI KIT | $
Materials for 6 sections
ADD-ON | $
Consumables for 1 class
C.4 Chemical Reactions in our World
15 lessons | 33 days of instruction
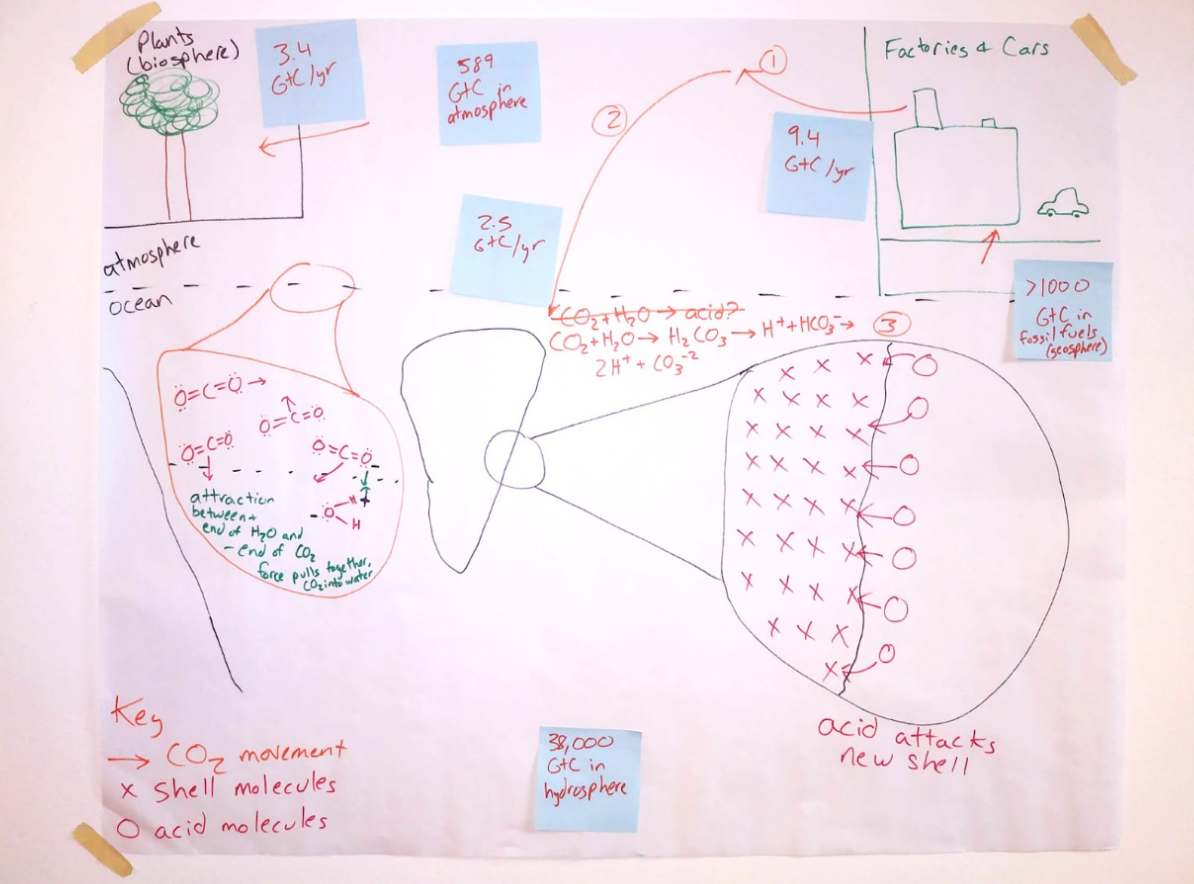
Why are oysters dying, and how can we use chemistry to protect them? This unit is designed to build a deeper understanding about chemical reactions by exploring reversible reactions through exploration of ocean acidification. Students watch case videos, analyze data, and read about how movement of carbon dioxide from the atmosphere to the ocean makes the ocean more acidic. They consider how oyster die-offs may affect communities that rely on oysters for a food source. Students break down this large scale problem into a few key subproblems so they can use chemistry to try to solve them. They figure out how changes in concentration of H+ ions in water leads to changes in water pH. They use their knowledge of chemical reactions and mathematical thinking (stoichiometry) to determine the amounts of a substance they could use to neutralize acidic water. Students consider engineering trade-offs, criteria, and constraints to use chemistry to develop a design solution at a specific site to address oyster die-offs. They apply their thinking in a culminating task around increasing rates of ammonia fertilizer.
Learn more in the Unit Storyline
SINGLE KIT | $
All materials for 1 class
MULTI KIT | $
Materials for 6 sections
ADD-ON | $
Consumables for 1 class
C.5 Energy from Chemical & Nuclear Processes
15 lessons | 31 days of instruction
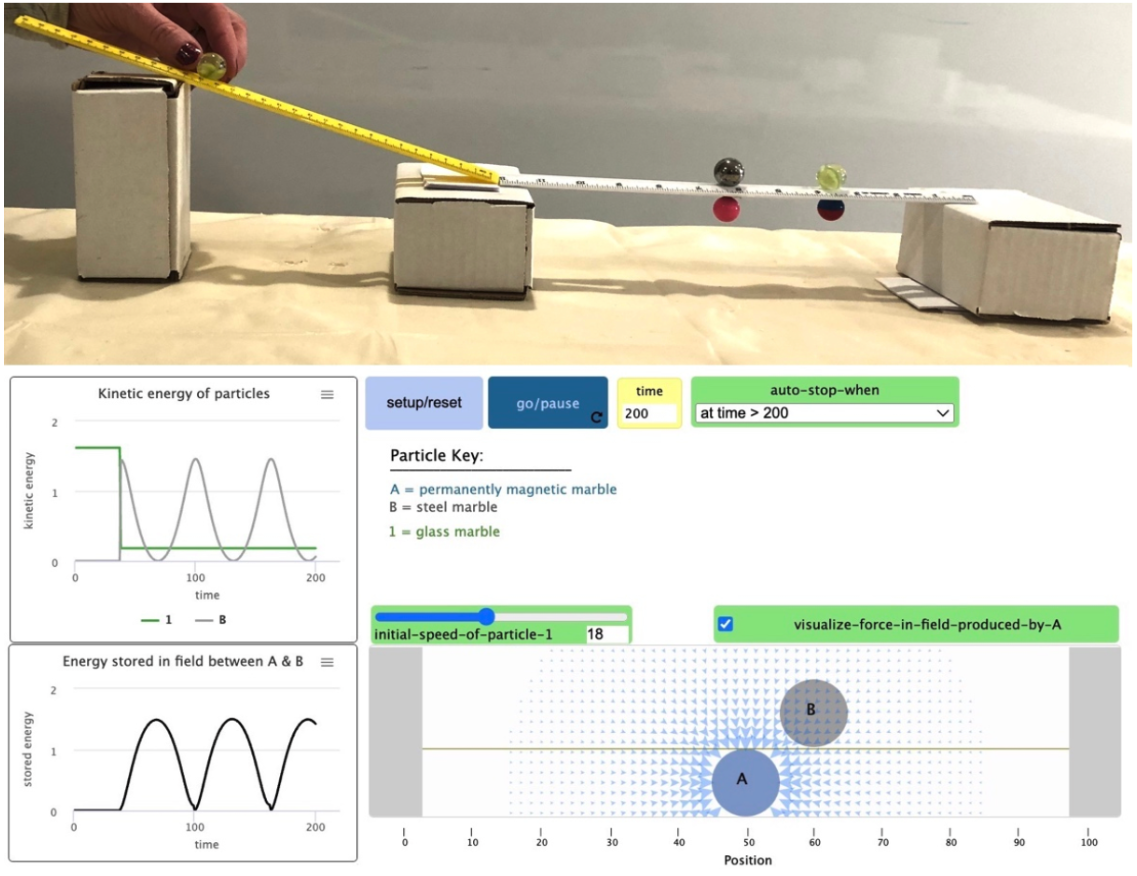
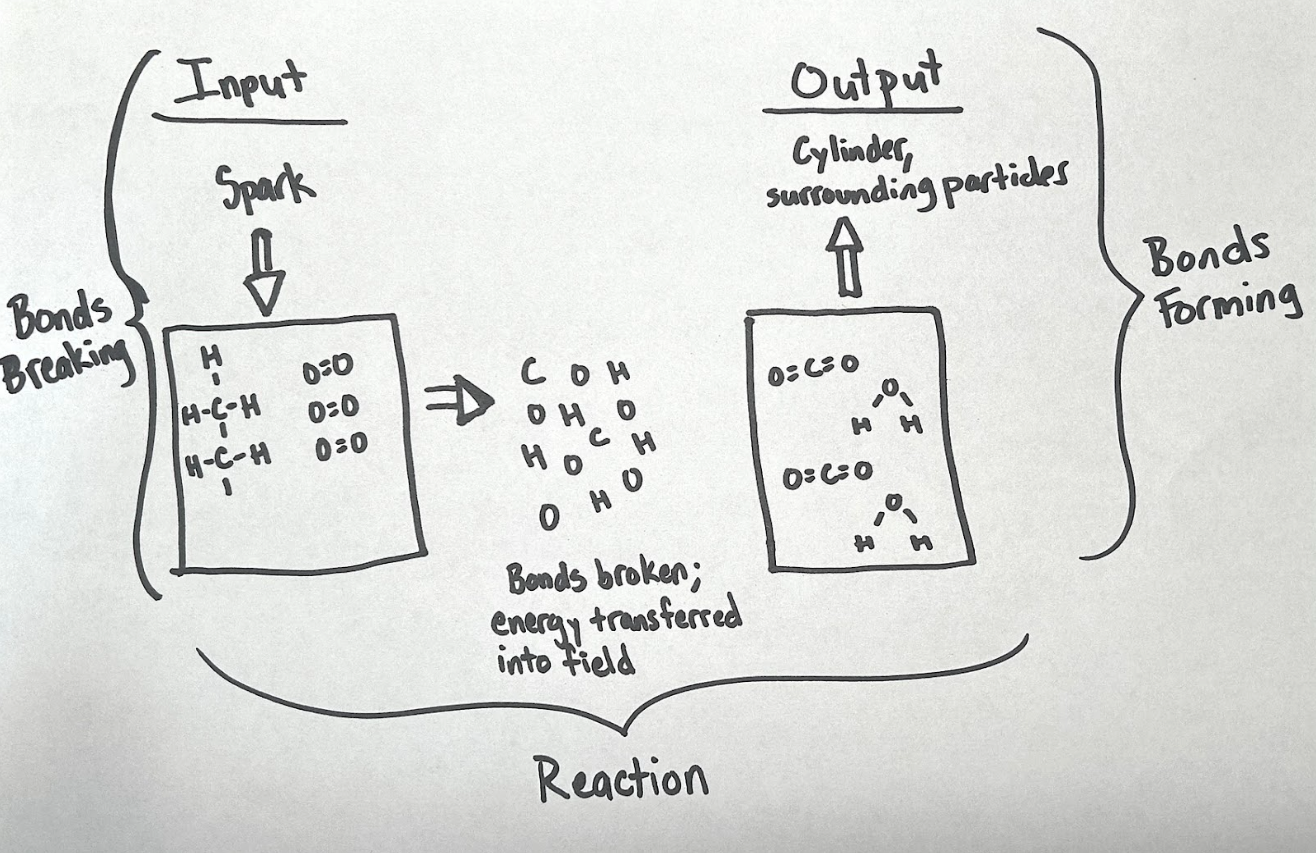
How can chemistry help us evaluate fuels and transportation options to benefit the Earth and our communities? This unit is designed to help students figure out ways to address climate change, first introduced in the first unit of the course. Students engage with information about different fuels used for transportation. They figure out what is happening in combustion reactions in gasoline, diesel, and biofuel engines, but are unsure where the energy is actually coming from. These use magnet marbles and simulations to figure it out and eventually quantify how much energy is released. They shift their focus toward engineering considerations and consider fuel options that are not carbon-based: electric vehicles, hydrogen, and uranium. They conclude the unit by evaluating a variety of fuels and other transportation solutions for a specific transportation goal. In doing so they develop nuanced arguments for a mix of transportation options to help address environmental, safety, and other concerns.
Learn more in the Unit Storyline
SINGLE KIT | $
All materials for 1 class
MULTI KIT | $
Materials for 6 sections
ADD-ON | $
Consumables for 1 class
Einstein Project is a 501(c)(3) non-profit STEM resource center driven to empower educators and inspire learners.